The processes of primitive streak formation and fate specification in the mammalian epiblast rely on feedback interactions between biochemical signals (morphogens and their inhibitors/activators) and tissue organization (epithelial cell polarity, adhesion and cytoskeletal tension). As these studies are difficult to perform in embryos, human pluripotent stem cells, considered as the equivalent of the epiblast, have become a reference system to dissect functional interactions and hierarchies.
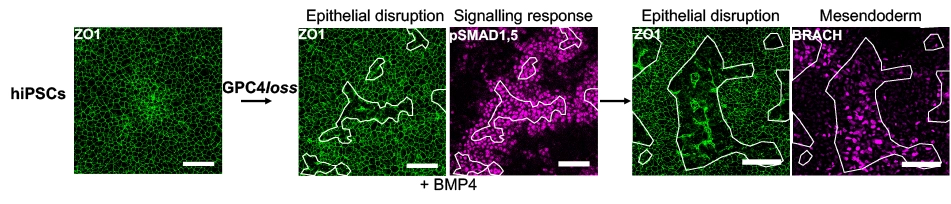
In a study published in Nature Communications led by Rosanna Dono in the Maina team, Thomas Legier, Diane Rattier and colleagues investigated the lineage commitment properties of hiPSCs down-regulated for the morphogen regulator GLYPICAN-4, in which defects in tight junctions (TJs) lead to areas of disrupted epithelial integrity. Using a combination of different hiPSC culture systems, this study shows that cells within disrupted areas become competent to sense the gastrulation signals, BMP4 and ACTIVIN A, and thus differentiate into mesendoderm. Furthermore, a protracted disruption of epithelial integrity sustains activation of the BMP4 and ACTIVIN A signaling pathway over time, affecting the efficiency and robustness of cell fate acquisition.
Collectively, these results reveal epithelial integrity as a key determinant of gastrulation signal activity and highlight a new mechanism guiding morphogen sensing and spatial cell fate change within an epithelium. Additionally, these findings highlight hiPSCs with reduced GLYPICAN-4 protein levels as a means to obtain new insights into developmental processes and for advancing into regenerative medicine.
It is possible that this mechanism of modulating epithelial integrity and signaling activity by altering TJs will be of more general relevance to other physiological and pathological events where there is a high degree of epithelial cell plasticity, such as epithelial barrier remodeling, tissue inflammation and tumorigenesis.
To know more
Legier, T., Rattier, D., Llewellyn, J. et al.
Epithelial disruption drives mesendoderm differentiation in human pluripotent stem cells by enabling TGF-β protein sensing.
Nat Commun 14, 349 (2023). https://doi.org/10.1038/s41467-023-35965-8