 Eukaryotes live in close association with the microorganisms that proliferate in the environment. While some, such as commensal bacteria that colonize the intestines of animals, can be beneficial, others are pathogenic and potentially dangerous to the host. As a result, animals have developed, over the course of evolution, strategies that limit the risk of infection with potentially harmful microbes. If this first step fails, the infected host activates an immune response that eradicates the pathogen. There is growing evidence to suggest that, in parallel with this “classical” immunity, the infected host modifies its behaviour to limit as much as possible the consequences of this infection on itself or on its offspring. This immunity, called behavioural immunity, requires a dialogue between infectious agents and the host’s nervous system.
Eukaryotes live in close association with the microorganisms that proliferate in the environment. While some, such as commensal bacteria that colonize the intestines of animals, can be beneficial, others are pathogenic and potentially dangerous to the host. As a result, animals have developed, over the course of evolution, strategies that limit the risk of infection with potentially harmful microbes. If this first step fails, the infected host activates an immune response that eradicates the pathogen. There is growing evidence to suggest that, in parallel with this “classical” immunity, the infected host modifies its behaviour to limit as much as possible the consequences of this infection on itself or on its offspring. This immunity, called behavioural immunity, requires a dialogue between infectious agents and the host’s nervous system.
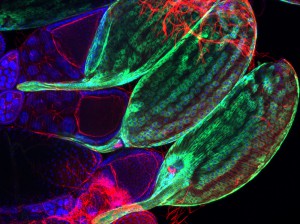
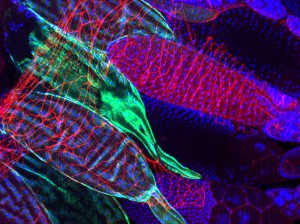 Julien Royet’s team uses the genetic tools generated in the fruit fly to dissect the molecular interactions between bacteria and the host’s nervous system. In an article whose main authors are Ambra Masuzzo and Léopold Kurz, the team demonstrates that the direct detection of a bacterial wall component called peptidoglycan, by only two octopaminergic neurons out of the approximately 100,000 contained in the fly’s brain, is sufficient to modify the behaviour of infected female. By inhibiting the activity of these two neurons, peptidoglycan reduces egg deposition by infected flies, allowing them to allocate more energy to pathogen elimination.
Julien Royet’s team uses the genetic tools generated in the fruit fly to dissect the molecular interactions between bacteria and the host’s nervous system. In an article whose main authors are Ambra Masuzzo and Léopold Kurz, the team demonstrates that the direct detection of a bacterial wall component called peptidoglycan, by only two octopaminergic neurons out of the approximately 100,000 contained in the fly’s brain, is sufficient to modify the behaviour of infected female. By inhibiting the activity of these two neurons, peptidoglycan reduces egg deposition by infected flies, allowing them to allocate more energy to pathogen elimination.
To know more :
-
Peptidoglycan-dependent NF-κB activation in a small subset of brain octopaminergic neurons controls female oviposition.
Ambra Masuzzo, Gérard Manière, Annelise Viallat-Lieutaud, Émilie Avazeri, Olivier Zugasti, Yaël Grosjean, C Léopold Kurz, Julien Royet
-
CNRS website



