Project title: Investigating the mechanobiology of self-organisation in human iPSC-derived skeletal muscles
Type of rotation: M1 (2 months) or M2 (6 months)
Supervisors: Qiyan MAO and Frank SCHNORRER
Introduction:
Both a force-generation machine and a major metabolic organ, skeletal muscles are of vital importance to the human body. Our team aims to understand how the muscle machine is built via self-organisation and mechanical feedback. Recently, using human iPSC-derived myogenic cultures, we revealed a striking capacity of human skeletal myofibers to self-organise into muscle bundles containing contractile sarcomeres and stable attachment foci, which allow tension level to rises on individual myofibers. We suggest that mechanical tension itself is key to coordinate the multi-scale self-organisation in human muscles (Mao, Acharya et al. 2022).
Project:
In this master project, you will investigate several potential mechanisms underlying tension build-up during human skeletal muscles self-organisation. You will perform cell culture with human iPSC-derived myoblasts, then use chemical inhibitors to perturb sarcomeric molecules such as actin and myosin, and attachment molecules such as integrin. You will further evaluate their impact on muscle self-organisation with customized image analysis scripts (Aim 1). In parallel, you will learn to build hydrogel substrates with varying rigidity, and perform Traction Force Microscopy to quantify tension build-up (Aim 2 – M2 only). The successful completion of the project will allow you to answer how mechanical tension is generated inside human myofibers by integrating intracellular molecular forces and extracellular mechanical environment. It will also grant you competence in human cell culture, mechanobiology and image analysis.
Competence to acquire at the end of the stage:
Human iPSC cell culture
Tunable hydrogel substrates [M2 only]
Laser-scanning confocal microscopy
Traction Force Microscopy [M2 only]
Quantitative image analysis with R
Reference:
Mao*, Acharya … Schnorrer*, Tension-driven multi-scale self-organisation in human iPSC-derived muscle fibers. Elife 11, 1–32 (2022).
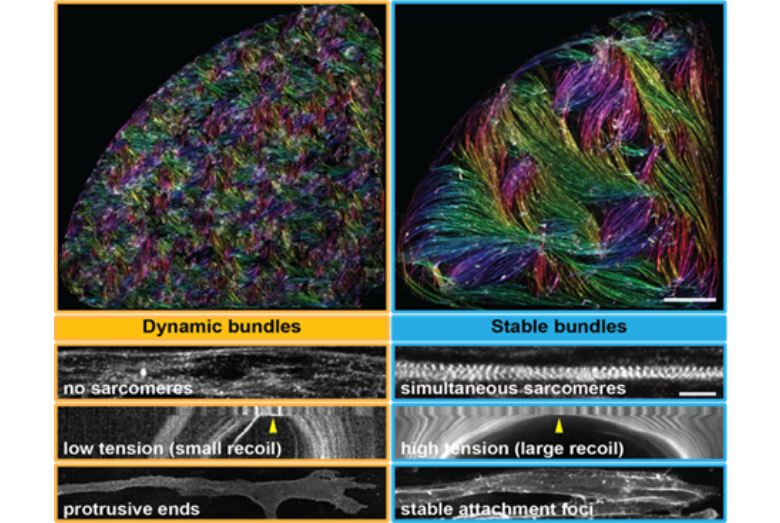
Figure 1: Human iPSC derived 2D muscle bundles self-organise into bundles, which spontaneously transit from dynamically merging bundles (left) to stably attached bundles (right) upon the formation of sarcomeres (titin immunofluorescence) and attachment foci (integrin β1 immunofluorescence). Tension increases dramatically at the transition point (kymograph of laser ablation of myofibers labelled with titin-GFP). Scar bars: 1 mm (upper), 10 µm (lower).